
Plastic Bottles Aren’t Waste Anymore; Scientists Turned Them Into Industrial Chemicals
Every year, millions of tons of plastic bottles are thrown away into landfills, and used vehicle batteries are being disposed of as hazardous waste. The average person thinks that these items are environmental hazards with little or no value left in them. However, there is increasing evidence that yesterday’s trash can be turned into tomorrow’s resource. Through a recent innovation in sustainable practices, researchers have developed a method of using only sunlight to convert discarded plastics and used battery acid into high-value chemicals for use in the production of dyes, coatings, pharmaceuticals, and other chemical processing.
Scientists at Cambridge University have developed a green chemistry method for converting plastic waste and used battery acid into useful chemicals, which are used for the manufacture of pharmaceuticals, dyes, coatings, and many other types of industrial applications. This technology provides a cleaner and more sustainable alternative to traditional methods of producing chemicals that are generally based on fossil fuels and consume significant amounts of energy during production.
In this study, the researchers focused their attention on the problem of hard-to-recycle plastics, including PET water bottles, nylon, and polyurethane foam. Instead of allowing these materials to accumulate in landfills or be incinerated, scientists developed a way to decompose them and use them as part of a chemical reaction that produces important chemicals called “anilines.” Anilines are commonly used in the chemical industry and are key building blocks for pharmaceuticals, plastics, agricultural products, and synthetic dyes.
The process utilizes a unique method of transforming the plastic into an industrially useful product using a method known as “photocatalytic transfer hydrogenation” (PTH). Although the terminology may sound technical, the basic principle behind this method is simple: using sunlight to help transfer hydrogen from the plastic-derived chemicals to another specific chemical, which will convert the chemical into an industrially useful product without using direct fossil-fuel-generated hydrogen gas.
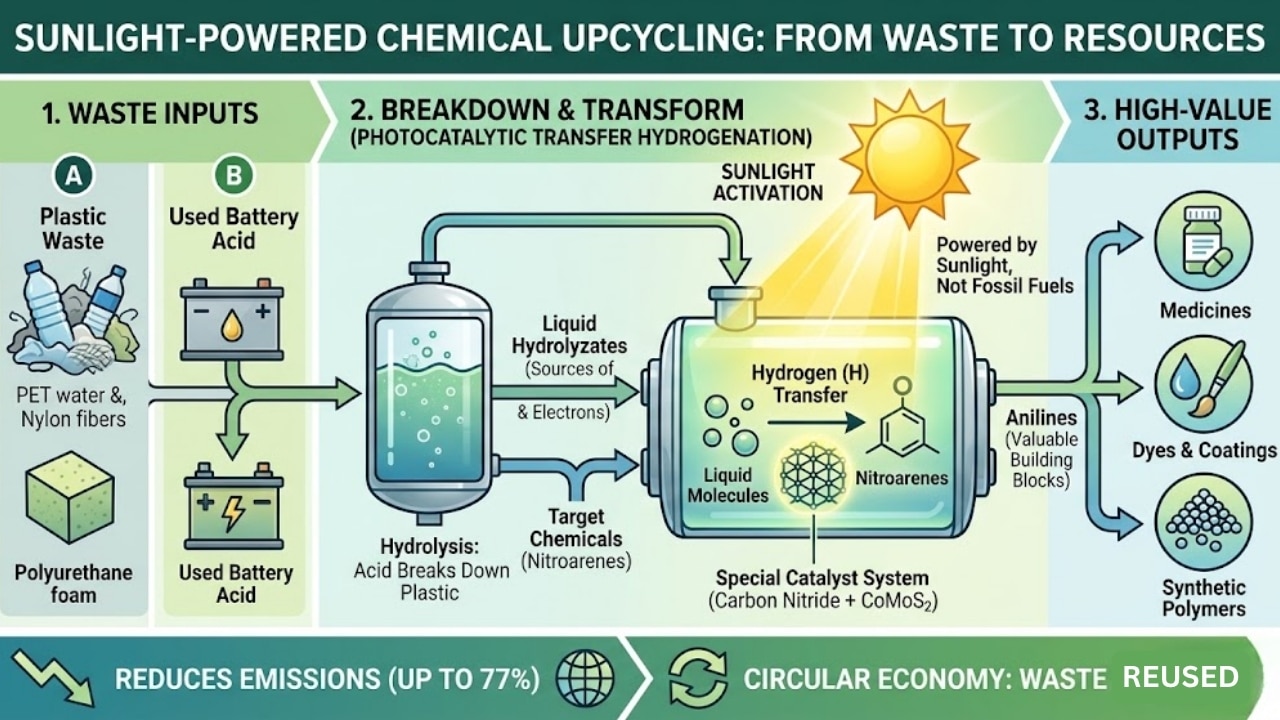
Another interesting part of this study is that the researchers used sulfuric acid that was recovered from old lead-acid batteries to break down solid plastics into liquid mixtures that are referred to as hydrolyzates. The hydrolyzates then provided the necessary protons and electrons required for the chemical transformation process.
In order for the process to be effective, a specialized catalyst system made of cobalt molybdenum sulfide (CoMoS₂) and carbon nitride was developed by a team member. The carbon nitride captures solar energy, and CoMoS₂ directs the chemical reaction to create the desired product. When combined, this state-of-the-art catalyst system can transform 24 different types of nitro-arene compounds to aniline with yields between 83% and 99%.
Most commercial manufacturing of these types of compounds takes place under extremely high-pressure conditions and high temperatures and necessitates the use of an expensive industrial infrastructure, whereas this environmentally friendly approach utilizes much milder process conditions to convert waste into value instead of creating value from new raw materials.
From an environmental standpoint, researchers estimate that this process could result in a reduction of carbon dioxide generation by 77 percent versus existing production methods. This would also help mitigate two large-scale, global waste issues, plastic waste and spent batteries, using a single integrated processing plant.
This type of discovery demonstrates how the potential for circular chemical manufacturing is increasing, that is to say, using waste materials over and over again to produce a new product rather than simply discarding those items. Scientists believe that this technology has the potential to be a major contributor to building a cleaner and more efficient future using renewable energy and sustainable chemistry.














































