CSIR NET Organic Chemistry Notes
Organic Named Reactions is an important topic under UNIT 6 of CSIR NET Chemical science syllabus – Common named reactions and rearrangements – applications in organic synthesis. Download PDF Notes for CSIR NET Chemical science organic chemistry – Organic Named Reactions below.
Weightage in the exam – This topic has about 10% weightage in the CSIR NET chemical science examination. Every year at least two questions are asked in Part B and 3-4 questions in Part C of the question paper.
To be through with For Organic Named Reactions topic, you can refer to the below-mentioned reference books:
- Advanced organic chemistry by Clayden Warren
- Organic chemistry by Peter sykes
- Organic chemistry by Jerry March
CSIR NET Organic Chemistry – Organic Named Reactions Notes
Introduction
An organic reaction mechanism is a complete, step-by-step account of how a reaction of organic compounds takes place. The description of an organic reaction mechanism typically includes designation of the overall reaction type (such as substitution, addition, elimination, oxidation, reduction, or rearrangement), the presence of any reactive intermediates, the nature of the reagent that initiates the reaction, the presence of any catalysis, and its stereochemistry.
Experiments are designed to produce results that provide logical evidence for (but can never unequivocally prove) a mechanism. For most organic reactions, some mechanisms are considered well established (that is, plausible) based on bodies of experimental evidence. Nevertheless, new data often become available that provide further insight into new details of a mechanism or that occasionally require a complete revision of an accepted mechanism. A common method for illustrating the progress of a reaction is the potential energy diagram, in which the system’s free energy is plotted as a function of the completion of the reaction.
Download Organic Named Reaction Complete Notes PDF
Reactions of organic compounds can be organized broadly in two ways,
- What types of reactions and
- How do these reactions occur
Overview of Types of Organic Reactions
The types of reactions organic compounds undergo are divided into four categories; these are
- Addition reactions
- Substitution reactions
- Elimination reactions
- Rearrangement reactions
Addition Reactions:
An addition reaction is a chemical reaction wherein two or more reactants come together to form a larger single product.
Addition reactions occur when two starting materials combine to form only one product with no atoms. In an addition, all parts of the adding reagent appear in the product; two molecules become one.
An addition reaction is being given by those compounds that have at least one pi-bond in them.
FREE Download – CSIR NET Physical Chemistry – Thermodynamics Notes PDF
For example,
Addition reactions can be classified into 3 categories based on the nature of initiating species.
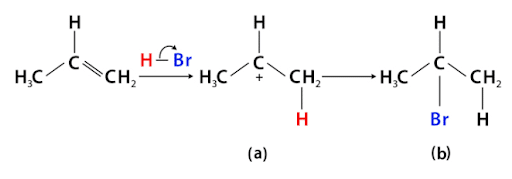
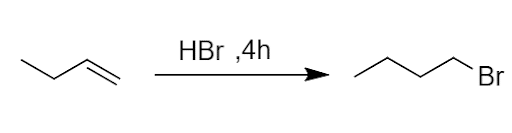
- Electrophilic addition: The alkenes and alkynes mainly give the reaction. Unsymmetrical alkenes and alkynes give addition reactions with unsymmetrical reagents according to Markovnikov’s rule.“ The Markovnikov Rule explains that in addition to reactions of alkenes or alkynes, the proton is added to the carbon atom with the highest number of hydrogen atoms.”
Some other substituted alkenes containing the -I group(-CX3, -CHO, -COOH, -NO2, etc) will follow antimarkovnikov’s rule.
- Nucleophilic addition: A nucleophile addition is an addition reaction in which a nucleophile adds to a pi bond of a compound. As a result, two new sigma bonds are formed.
- Free radical addition: The free radical reaction mainly involves the free radical species in the reaction. This reaction mainly follows both markovnikov and antimarkovnikov’s rule.
The mechanism of this reaction is the same as for most radical reactions. In the presence of radical, anti-Markovnikov hydroamination simply outpaces the regular addition pathway, which consists of the following steps: Initiation, propagation, and termination.
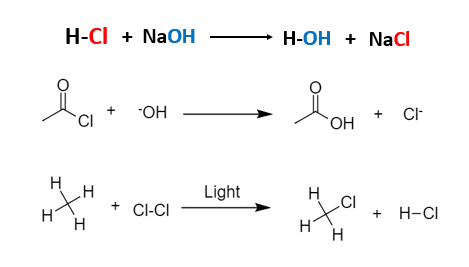
- Substitution Reactions:
A substitution reaction is a type of chemical reaction where an atom or functional group of a molecule is replaced by another atom or functional group.
Substitution reactions occur when two starting materials exchange groups to form two new products. In a substitution, one group replaces another.
Example,
A Substitution reaction can be classified according to the nature of the substituent. As follows,
Free radical substitution: CONTD…
Download Organic Named Reaction Complete Notes PDF
Aazad Batch 2022 For CSIR NET Chemical Science (12 months)
👉 Grab FREE Bioinformatics Internship with this Course




































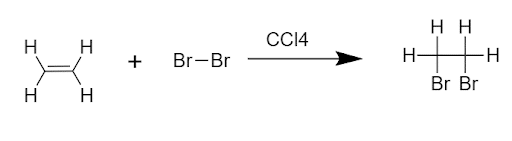
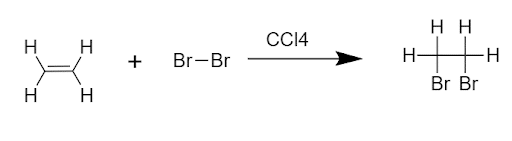













Super