What is Clinical Pharmacology?
Understanding The Basics Of Clinical Pharmacology
The study of drugs in humans is termed as Clinical Pharmacology. It is supported by the basic science of pharmacology, with the added focus on the application of pharmacological principles and methods in the real world. Clinical pharmacology has a broad scope, from the discovery of new target molecules to the effects of drug usage in whole populations. Clinical pharmacology is important for healthcare professionals because it is the scientific discipline that supports the rational prescribing of medicines in order to prevent future disease, treat illness, and alleviate symptoms.
Let’s now dive deep into clinical pharmacology to understand:
- How drugs are developed and where clinical pharmacology studies are performed.
- Pharmacokinetic Studies: Evaluates the contribution of absorption, distribution, metabolism, and elimination to the drug concentration-time curve.
- Pharmacodynamic Studies: Explains key terms such as dose-response relationships, including receptors and actions of drug targeting.
- Predict how individual variability in the Pharmacokinetics affect the pharmacodynamics such as efficacy and toxicity.
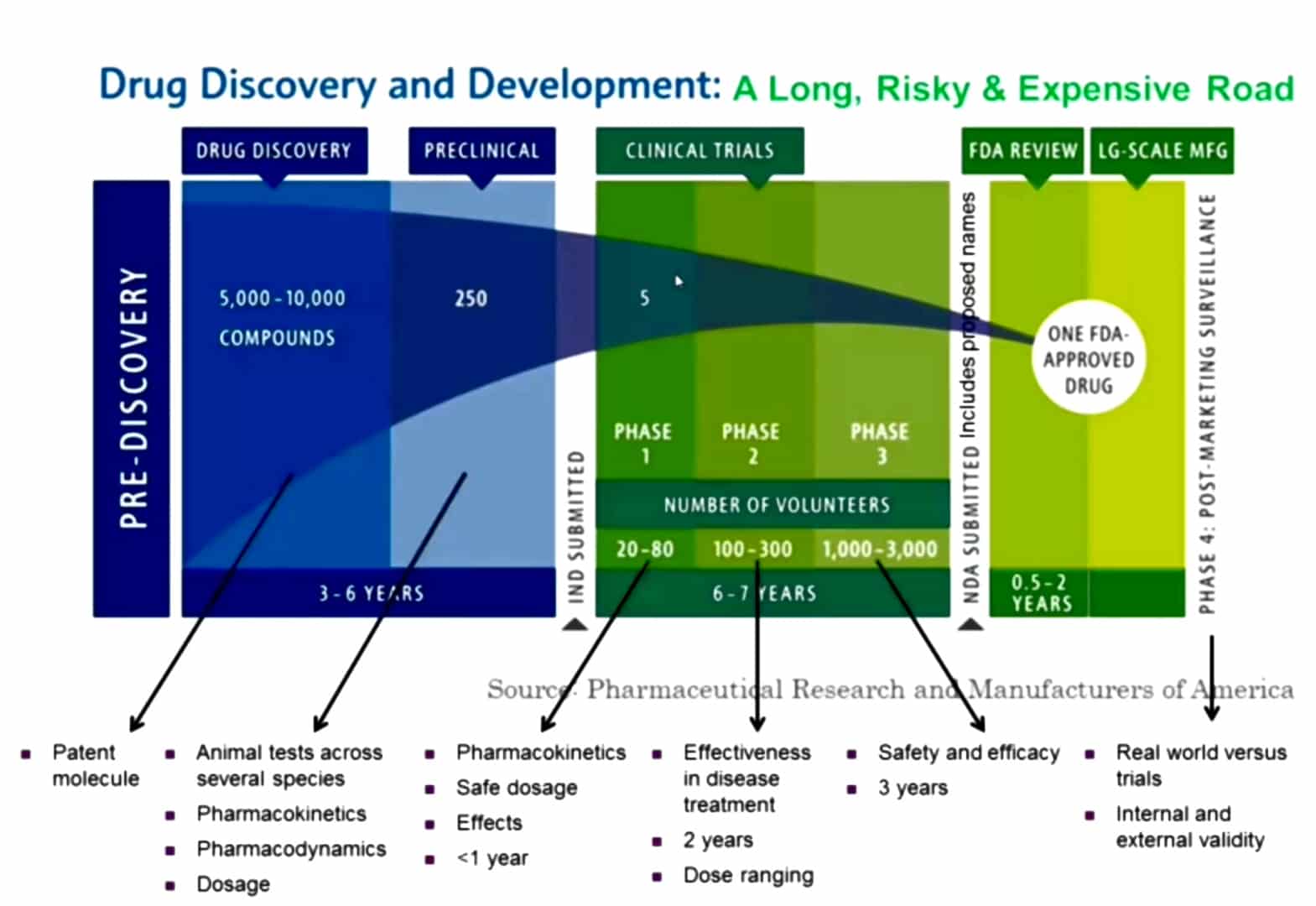
This image depicts the process of drug discovery and development in the different phases of drug development. The pre-discovery phase is the identification of molecules, which includes the early drug development and then the
preclinical drug development involving pharmacology studies in animals. A lot of the studies that we perform in clinical pharmacology that are involved in patients as part of clinical trials are also performed in preclinical studies. The clinical pharmacology studies are performed in phase 1 and phase 2 studies of drugs during development. Sometimes in phase 3 which are a much larger study and then also in post-approval studies which are called in many cases phase 4 studies.Clinical Pharmacology has Two Basic Parts:
- Pharmacokinetics
- Pharmacodynamics
Pharmacokinetics is what the body does to the drug, how the body handles the drug, clears it, distributes, and other factors. Pharmacodynamics is what the drug does to the body, the effects of the drug on the body such as for efficacy, targeting, and toxicity.
Pharmacokinetics
In pharmacokinetics, we can explain the pharmacology of the drug mathematically. It is the drug’s journey through the body and how the drug is handled by the body. There are four different basic processes (ADME) to pharmacokinetics:
- Absorption
- Distribution
- Metabolism
- Elimination
When a drug is dosed either orally or IV(intravenous), it goes into the central compartment, which is the absorption phase, then it goes into the peripheral compartment which is a distribution phase, and then lastly the drug is eliminated which is the elimination phase.
How pharmacokinetics studies are performed for various drugs and development?
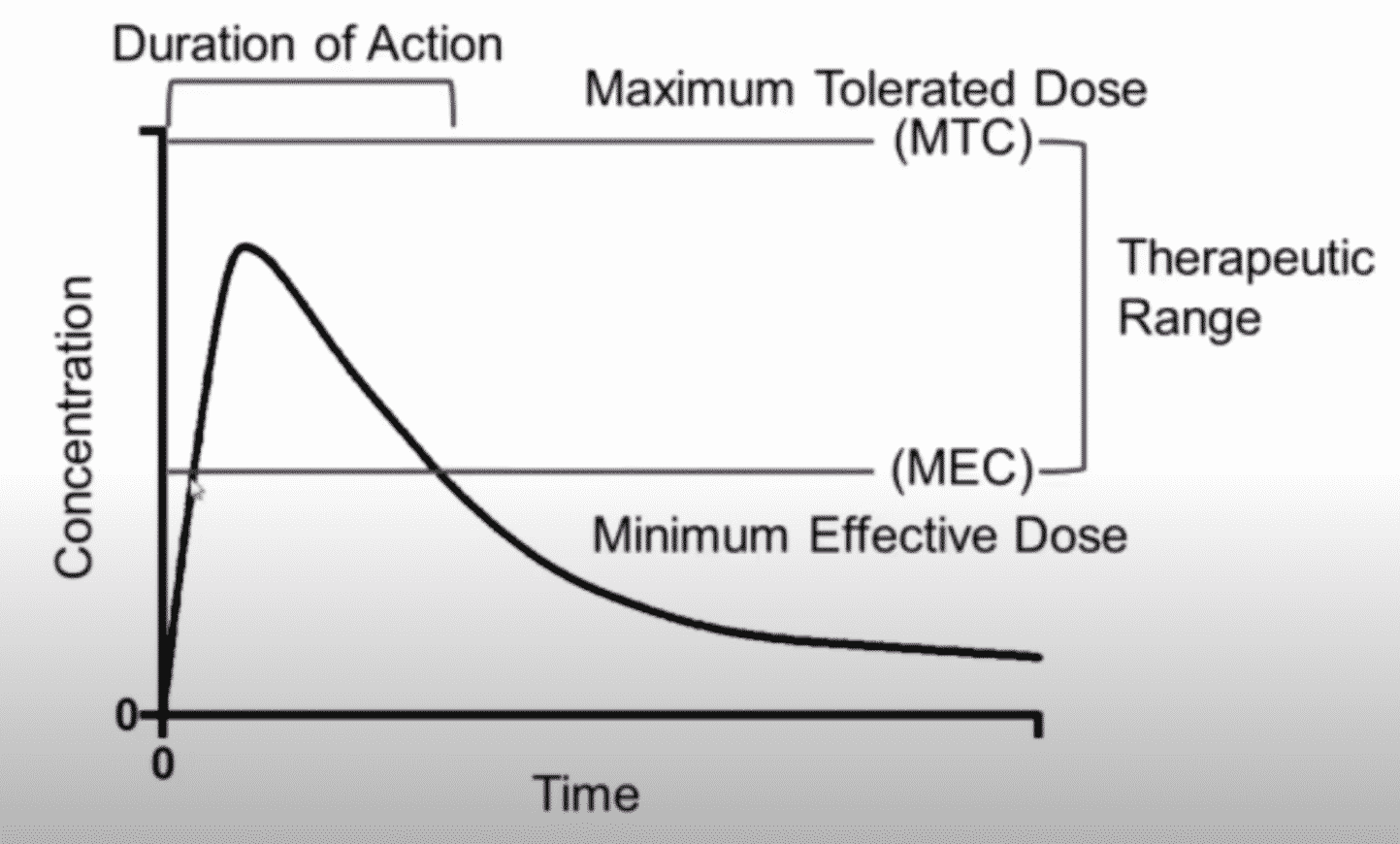
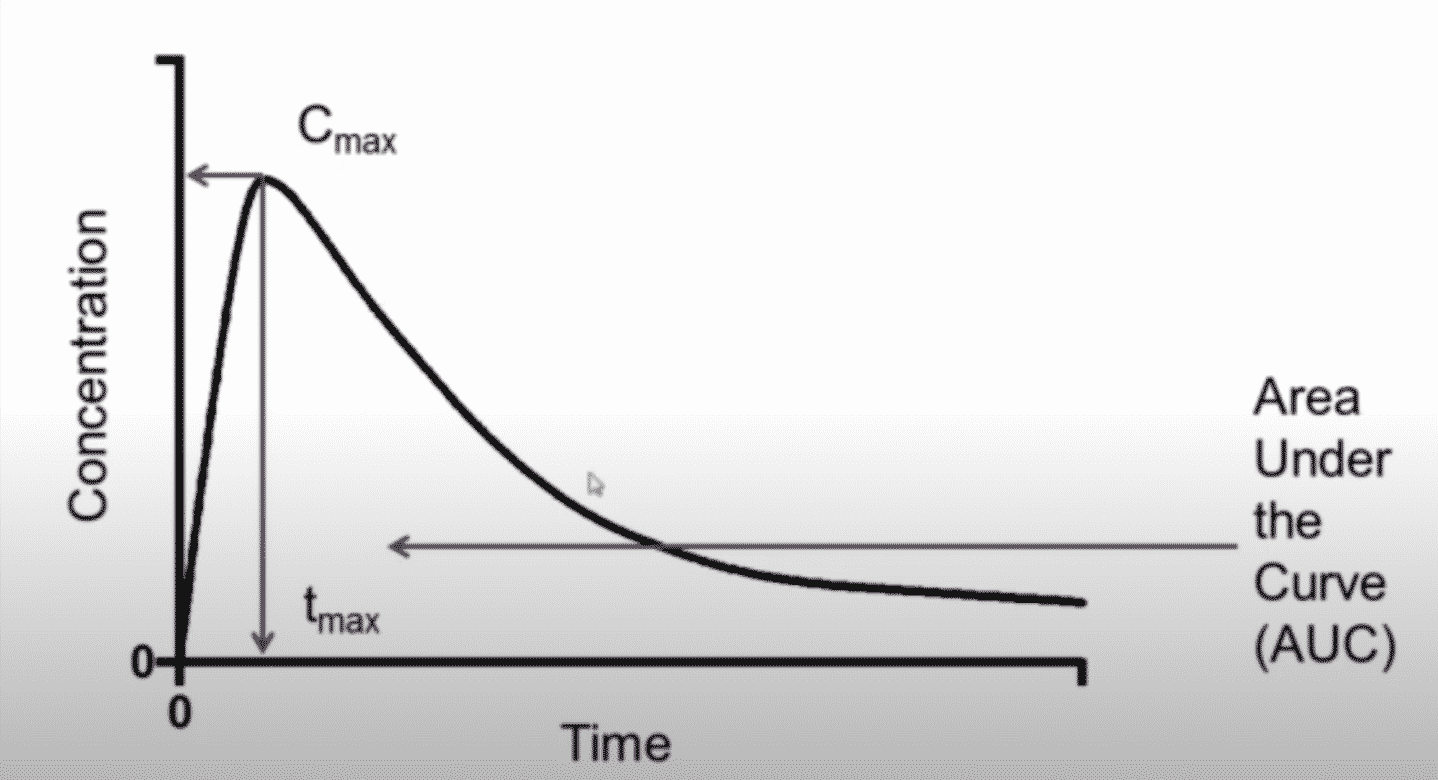
This graph depicts the concentration versus time curve which is involved in the pharmacokinetic studies.
The graph has time on the X-axis, and concentration on the Y-axis, includes terms such as the minimum effective dose or exposure and the maximum tolerated dose, therapeutic range, and important pharmacokinetic terms such as the C-max or maximum concentration, t-Max or the time of the maximum concentration and an area under the concentration-time curve which is the AUC in a measure of overall exposure.
In these studies, we try to evaluate these different pharmacokinetic parameters and eventually see how they predict the pharmacodynamic response.
Routes of Administration
Drugs can be administered through various routes of administration such as:
- Parenteral administration (Example: intravenous(IV), intramuscular(IM), or subcutaneous injection). Most drugs that use a parenteral administration are via IV.
- Oral administration: (Example: tablets, capsules, suspension, or liquid)
- Sublingual administration: (Example: tablets)
- Local administration: (Example: ointment, cream, patch, suppository, or inhalation)
Bioavailability
Bioavailability is a very important pharmacokinetic term. It is the fraction or percentage of a drug that reaches the systemic circulation, or blood exposure. For instance, if you give a dose orally, it goes in and dissolves or breaks down into the gut that is then absorbed into the blood and is metabolized by the liver through, and ultimately what gets to the blood after the liver is what is bioavailable. Bioavailability is influenced by absorption and metabolism.
Factors affecting absorption distribution:
- Factors affecting absorption
- Tissue permeability
- Blood flow
- Binding to plasma proteins
- Binding to additional cellular compartments
All these factors determine where the drug and how fast the drug distributes throughout the body.
Protein binding
Protein binding is also a very important pharmacokinetic term. It’s related to the binding of the drug to plasma proteins such as albumin, beta globulin, and alpha acid glycoprotein. It’s important to remember that drugs that are bound to these proteins have no effect. The amount of drug bound is determined by different concentrations such as the free drug concentration, the protein-bound concentration, and affinity for binding sites.
Pharmacokinetic Mechanism: Elimination
There are three different types of eliminations.
First is enzymatic metabolism where the goal is to enhance elimination from the body. The enzymatic metabolism mostly occurs in the liver by reactions that increase the water solubility. The metabolites are then secreted back into the blood or into the bow, where they’re eliminated from the body. There are different phases of enzymatic metabolism:
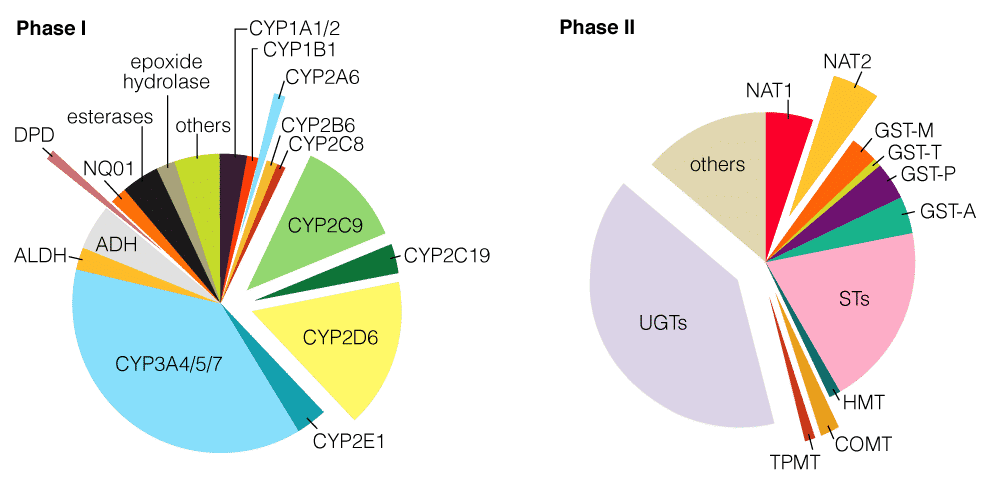
Phase I, which is making the drug more hydrophilic such as sip 450 enzymes in this case and then there’s the phase II metabolism which involves conjugating it to also make it more water-soluble, so that is eliminated.
The second type of elimination is renal elimination. There are two different types of renal elimination: Filtration, which goes through the renal glomerulus and its elimination is through the urine; Secretion, where the drugs are actively secreted through the renal tubules of certain drugs and again they go through in elimination through the kidney and out into the urine.
The last type of elimination is a relatively new or novel form of elimination. It’s a cellular elimination via the mononuclear phagocyte system or MPs system and this is for complex drugs such as nanoparticle conjugates and biologics, such as antibodies or antibody-drug conjugates.
Half-Life
An important pharmacokinetic parameter is a half-life. It’s defined as the time it takes for half the drug to be administered. Each drug has its own half-life that needs to be characterized. When you’re giving repeated doses of a drug, either if it’s an IV infusion and you stop the infusion, then the drug clears, which is termed as the washout period. The time it takes for half the drug to be eliminated is what we would call the half-life.
Pharmacodynamics
Pharmacodynamics is what the drug does to the body. It’s related to the drug’s destination or purpose. It involves efficacy and toxicity, and important terms such as therapeutic index, sites of action, and an affinity for receptors.
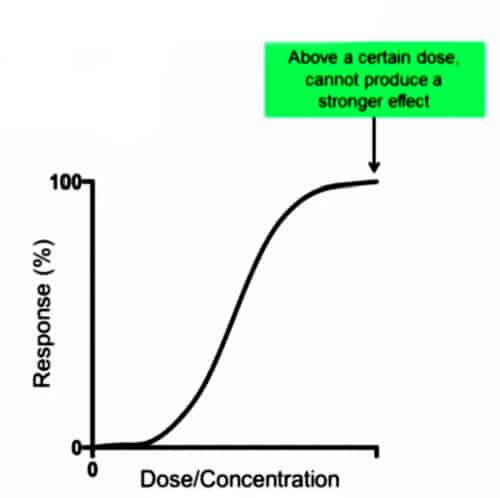
When you give a dose or a concentration of drug measured in pharmacokinetic studies, the degree of response goes from zero up to a hundred percent and you get this sigmoid or curve as shown in the graph. Once you reach a point where giving more drugs or a higher concentration of drugs, you get no more added effect. This would be the maximum effect that can occur. Above this point, not more should be added because there will no added response, and would just cause off-target effects or toxicity.
Potency
The potency is a pharmacodynamics term related to the relative strength of response for a given dose.
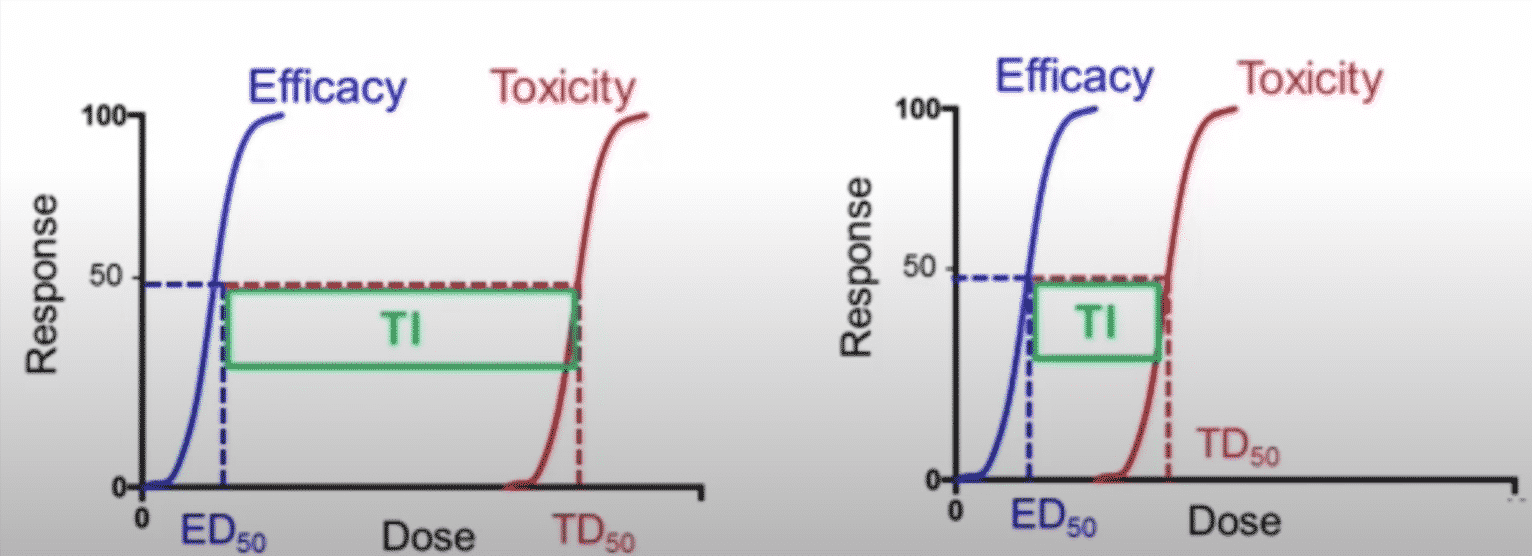
Therapeutic index
The therapeutic index is a very important pharmacodynamics term. The therapeutic index is related to the toxic or lethal dose at 50%.
An easier way to think about the therapeutic index is to look at the range or distance between what is required for efficacy or what is required for toxicity below:
The efficacy curve represented by the blue line, and the toxicity curve represented by the red line. The distance or interval, or exposure range between what causes efficacy and what causes toxicity is called the therapeutic index. The agent on the left would have a wider therapeutic index, which is a good parameter or a good characteristic of the drug. The agent on the right has a narrow therapeutic index, which means the exposure that causes or associated with efficacy or causes toxicity is very close, and this can be problematic for a particular drug, due to variability in kinetics and exposure from patient to patient and dynamic response.
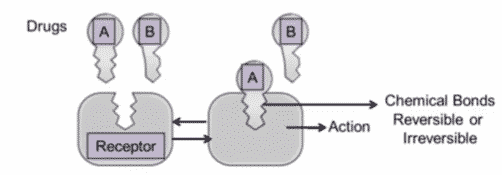
Molecular Mechanisms of Actions
There are different molecular mechanisms of actions in pharmacodynamics. Drugs must bind to a specific site to elicit a response called the drug-receptor or site of action. There’re many different targets such as lipids, nucleic acids, or proteins (most receptors). It’s a Lock-and-key analogy:
With a receptor, you need the drug, either drug a or drug B, to bind to the receptor to achieve a response. If a drug binds, it achieves a response or an action. The interactions are either reversible or irreversible. Multiple drugs bind to multiple different receptors to elicit a pharmacologic response.
Agonists and Antagonists
There are different types of interactions or agents, agonists, and antagonists. The therapeutic effects of drugs can be through agonists or antagonist activity. Drug interactions occur when agonists and antagonists are present together.
Agonists bind to the receptor and cause measurable effects. Agonists are driven by affinity and intrinsic activity. There are partial agonists that have an affinity and less intrinsic activity.
Antagonist binds to a receptor but no measurable or no measurable cellular or physiological change occurs. It blocks the usual receptor effect and it can reduce the effect of an agonist. They do have affinity but no intrinsic activity. The different antagonists can be competitive or they’re binding to the same site as the agonist or they can be overcome with higher concentrations. They can also be non-competitive.
Studies are ongoing for all drugs at different phases of development to understand how variability in pharmacokinetic parameters such as absorption, distribution, elimination, and the overall exposures affect the pharmacodynamic response, whether it makes the response steeper or less steep. Thus, these are important concepts that need to be performed for all drugs.
By studying the medication’s restorative effect to amplify the drug effect and reduce the side effects, clinical pharmacology promotes the rational use of medications in humans. Through development, research, and regulation of medications, clinical pharmacologists bridge the gap between science and the practice of medicine.
Source : NIH Clinical Center