Table of Contents
Enzyme Structure Explained: Functions, Types, Classification, and Chemistry
Have you ever wondered how your body turns your breakfast into energy or how your body knows it needs to heal a cut so quickly on its own? It is all because of a tiny protein called an enzyme. Their special and unique enzyme structure allows them to perform many essential enzyme functions, without which we would not be able to survive.
Enzymes are also known as biocatalysts as they help in biological processes such as breaking down food into energy, e.g, our saliva has an enzyme known as amylase that helps to break down starch from complex sugar to turn it to simple sugar called glucose. This Glucose will be transported in our body through the bloodstream, which will be absorbed by cells and produce energy. Enzymes are a crucial part of industries like drugs and medicine, food, and biofuel, so it is important to understand the enzyme chemistry, types of enzymes, and enzyme activity to unlock the secrets of life.
In this article , we will not only discuss the enzyme structure, but also give you a brief understanding of enzyme classification and enzyme mechanisms.
What is an Enzyme?
An enzyme is a biological molecule called a protein that acts as a catalyst for biochemical reactions inside our bodies. They help speed up reactions, and unlike other substances, enzymes are not entirely consumed in the process, so that they can be reused.
Enzymes play a crucial role in enabling essential biochemical reactions to occur efficiently and at the right speed to sustain life, such as helping in digesting food, synthesizing DNA, and generating energy from nutrients.
Essential enzyme functions, such as digesting food, producing energy, and building molecules that are essential for our body. Each enzyme is designed uniquely and works on a substance specifically, showing its enzyme activity and precision, like how a key would fit into a lock.
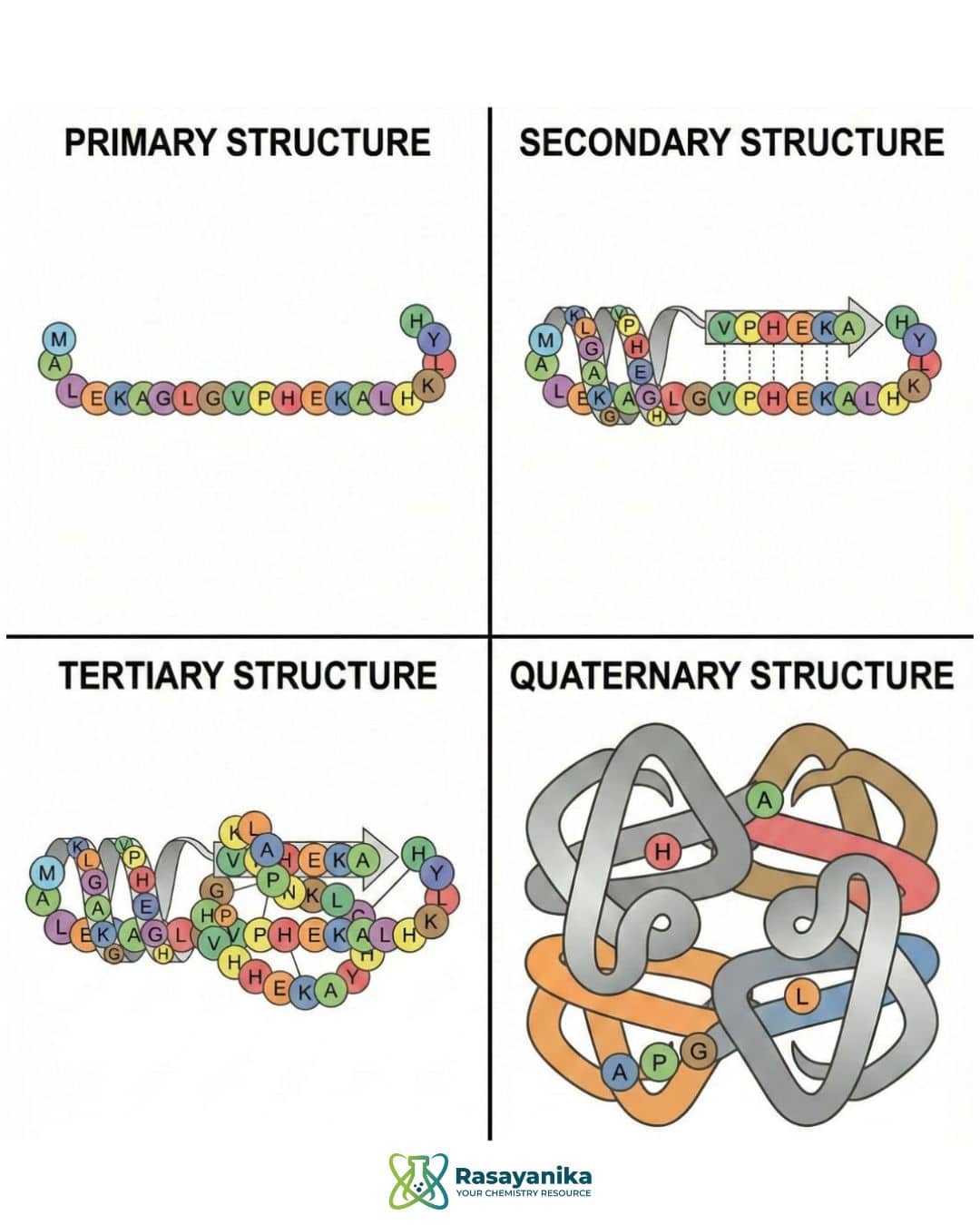
Enzyme Structure
The structure of an enzyme is essential for it to work and perform catalytic activity. Enzymes are proteins that can be described at four structural levels. The following are the four enzyme structures:
- Primary structure: A single sequence of amino acid chains.
- Secondary structure: The single sequence of amino acids will fold and make the chain into patterns like α-helices and β-sheets.
- Tertiary structure: It is a 3D folding of the primary and secondary structures, which gives it its unique shape.
- Quaternary structure: When a Tertiary structure folds further, it is called quaternary structure, and some enzymes have polypeptide chains working together.
All enzymes have a specific shape, which helps them to determine the active sites on a substrate. It is essential because only the right substrate should fit into the active sites, allowing the enzyme to carry out its activities efficiently. If there are any changes in the structure, then it will affect enzyme activity.
Enzyme Chemistry
It explains how enzymes help biochemical reactions to occur quickly and accurately. They bind to a specific molecule called a substrate to form a complex called an enzyme-substrate complex. This helps the reactions to occur soon by reducing the activation energy. Activation energy is the energy needed to start a chemical reaction.
Enzymes help in stabilizing the transition state, a short-lived form of substrate during the reaction, allowing it to convert into product at a fast speed. Some enzymes also require helper molecules such as the cofactors (often metal ions) or coenzymes (small organic molecules like vitamins to perform their function.
It is essential to understand the process of learning the enzyme mechanism, which explains how an enzyme catalyzes reactions step by step in our body or in industrial processes.
Types of Enzymes
Enzymes can be classified into different types depending on the following:
| Category | Type | Description / Example |
| Based on Substrate | Proteases | Act on proteins |
| Lipases | Act on fats | |
| Amylases | Act on starch | |
| Nucleases | Act on nucleic acids | |
| Based on Location | Intracellular enzymes | Work inside cells (e.g., metabolic enzymes) |
| Extracellular enzymes | Work outside cells (e.g., digestive enzymes) | |
| Based on Chemical Nature | Simple enzymes | Protein only |
| Conjugated enzymes | Protein + non-protein part | |
| Apoenzyme | Protein is part of an enzyme | |
| Cofactor / Coenzyme | Non-protein component | |
| Holoenzyme | Apoenzyme + cofactor (active form) | |
| Based on Stability | Thermostable enzymes | Active at high temperature (e.g., Taq polymerase) |
| Thermolabile enzymes | Destroyed by heat | |
| pH-stable enzymes | Active in extreme pH (e.g., pepsin) | |
| Based on Function | Digestive enzymes | Break down food (e.g., amylase) |
| Metabolic enzymes | Drive energy reactions (e.g., ATP synthase) | |
| Genetic enzymes | DNA/RNA processes (e.g., DNA polymerase) | |
| Detoxifying enzymes | Remove toxins (e.g., P450 enzymes) | |
| Signaling enzymes | Cell regulation (e.g., kinases) | |
| Repair enzymes | Fix DNA damage (e.g., DNA ligase) |
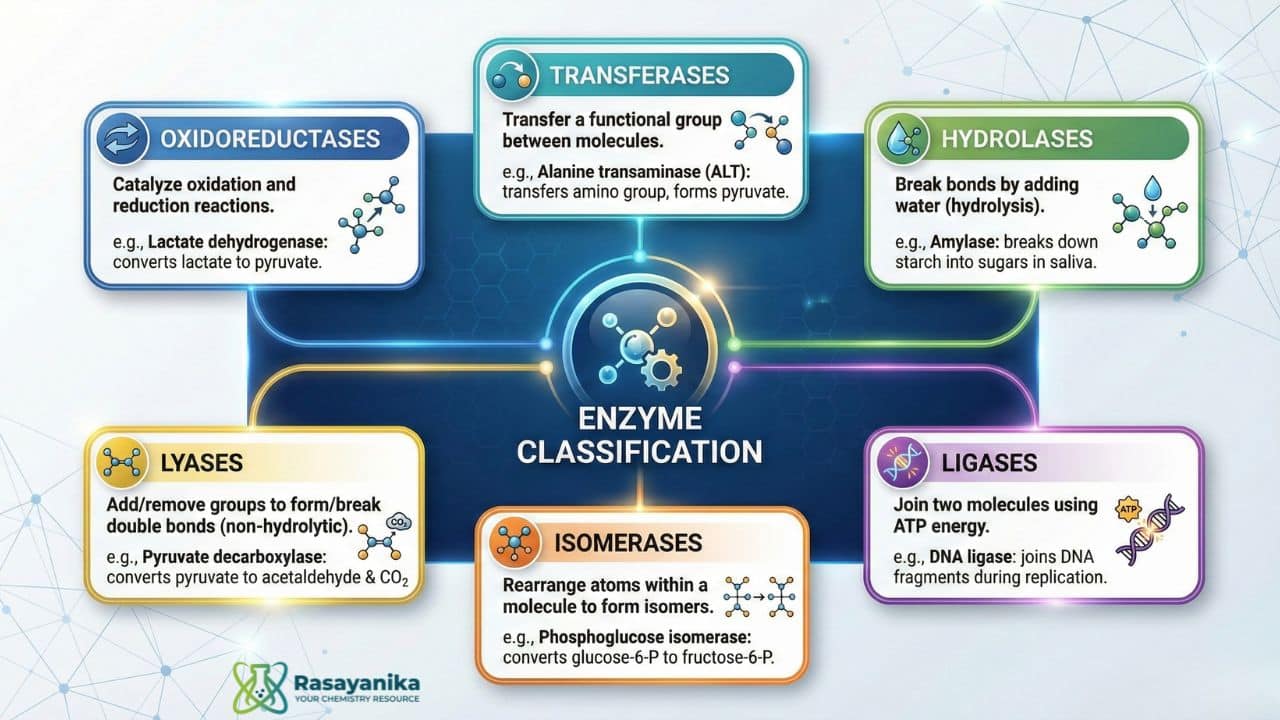
Enzyme Classification
There are six types of enzymes, and this classification is officially accepted and used worldwide based on the type of reaction they catalyze. The following are the main types of enzymes with examples:
- Oxidoreductases
These enzymes catalyze both oxidation and reduction reactions. E.g, Lactate dehydrogenase helps to convert lactate to pyruvate in cells.
- Transferases
These enzymes transfer a functional group from one molecule to another. E.g., Alanine transaminase (ALT) transfers the amino group of alanine to alpha-ketoglutarate, forming pyruvate and glutamate.
- Hydrolases
These enzymes will break bonds of molecules by adding water, and this is called hydrolysis. E.g, Amylase present in saliva will break down starch into sugars.
- Lyases
These enzymes will add or remove a group to break or add a double bond without hydrolysis. E.g, Pyruvate decarboxylase helps in fermentation by converting pyruvate into acetaldehyde and CO2
- Isomerases
These enzymes will rearrange atoms within a molecule to form isomers. They help molecules to switch between structural, geometric, or optical forms. E.g, Phosphoglucose isomerase converts glucose-6-phosphate to fructose-6-phosphate.
- Ligases
These enzymes help join two molecules together using energy released from ATP. E.g, DNA ligase joins fragments of DNA during replication.
Factors Affecting Enzyme Activity
The enzyme activity and enzyme function can be influenced by several factors:
- Temperature:
Enzymes work best at an optimal temperature. Too high or too low temperatures can slow down or denature the enzyme, e.g, Human enzymes work best around 37°C.
- pH:
Each enzyme has an optimal pH. Too acidic or too basic conditions can reduce activity, e.g, Pepsin in the stomach works best in acidic pH (~2), while trypsin in the small intestine works best in slightly basic pH (~8).
- Substrate concentration:
Increasing substrate levels generally increases enzyme activity, up to a point. Once all active sites are occupied, the reaction rate plateaus.
- Inhibitors and activators:
Inhibitors decrease enzyme activity.
- Reversible inhibition: Competitive inhibitors like methotrexate temporarily block the active site.
- Irreversible inhibition: Cyanide permanently disables enzymes involved in cellular respiration.
- Activators increase enzyme activity by helping the enzyme function more efficiently.
Understanding these factors is important for controlling enzyme function in both the body and industrial processes.
Mechanism of Enzyme Action
Enzymes speed up reactions through a simple but efficient enzyme mechanism:
- Substrate binding:
The substrate attaches to the enzyme’s active site, which is highly specific.
- Enzyme–substrate complex formation:
The enzyme holds the substrate in place, lowering the activation energy needed for the reaction.
- Conversion to product:
The enzyme carries out the necessary chemical changes, turning the substrate into a product.
- Product release:
The products leave the active site, and the enzyme is free to repeat the process.
Two models explain how enzymes bind substrates:
- Lock-and-key model: The active site is a perfect fit for the substrate.
- Induced fit model: The enzyme slightly changes shape to fit the substrate better (more accurate today).
Applications of Enzymes
Enzymes play an essential role beyond the human body, and their enzyme function and high enzyme activity make them valuable in many real-world fields.
Medical Applications
- Used in diagnostics, such as detecting glucose levels using glucose oxidase.
- Applied in therapeutics, for example, using proteases to dissolve blood clots or digestive enzyme supplements.
Industrial Applications
- In the food industry, enzymes help in brewing, cheese making, and improving bread quality.
- In biofuels, enzymes like cellulases break down plant material to produce ethanol.
- In detergents, enzymes remove stains by breaking down proteins, fats, and starch.
Research and Biotechnology
- Enzymes such as DNA polymerases are essential for PCR, genetic engineering, and DNA cloning.
- Used in biocatalysis to create environmentally friendly chemical reactions.
The enzyme structure plays a vital role in determining how it performs its specific function, as even small changes in structure can significantly affect enzyme activity. Understanding enzyme chemistry and the enzyme mechanism will explain how enzymes speed up life-sustaining reactions with remarkable precision. This knowledge has driven many innovations in medicine, industry, and biotechnology, from drug development to sustainable biofuels. Continued exploration of enzymes will further unlock their potential in applied sciences and future technological advancements.